Description: Participants: Interactions 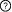 This is a sequential switch. The interactions in this switch take place in a defined order. The order is defined by numbers to the left of each interaction. More than one interaction can occur in each step, these interactions occur independently but are necessary for future step This is a sequential switch. The interactions in this switch take place in a defined order. The order is defined by numbers to the left of each interaction. More than one interaction can occur in each step, these interactions occur independently but are necessary for future step1Interaction #1 SMAD3 - PIN1 Is mutually exclusive with Interaction #4 SMAD3 - NEDD4L Interfaces (1) DOC_WW_Pin1_4 motif (176IPETPP181) in Mothers against decapentaplegic homolog 3 (SMAD3) (2) WW domain (7-37) in Peptidyl-prolyl cis-trans isomerase NIMA-interacting 1 (PIN1) Interaction Regulation PTM-dependent Induction (Phosphorylation of T179 on Mothers against decapentaplegic homolog 3 (SMAD3)) of the Mothers against decapentaplegic homolog 3 (SMAD3) DOC_WW_Pin1_4 motif - Peptidyl-prolyl cis-trans isomerase NIMA-interacting 1 (PIN1) WW domain interaction Regulatory Enzymes for switch Modifying enzymes for residue: T179: CDC2/CDKX subfamily Inferred Regulatory Enzymes for switch Putative modifying enzymes for residue: T179 : Cyclin-dependent kinase 2 (CDK2), Cyclin-dependent kinase 2 (Cdk2), Cyclin-dependent kinase 4 (Cdk4), Cyclin-dependent kinase 8 (CDK8), Cyclin-dependent kinase 9 (CDK9), Cyclin-dependent kinase 4 (CDK4), Mitogen-activated protein kinase 1 (MAPK1). Additional Information Affinity : T179: >100 µM, pT179: 12.6 µM Structural information: 2LB3 1Interaction #2 SMAD3 - GSK3B Interfaces (3) MOD_GSK3_1 motif (201DAGSPNLS208) in Mothers against decapentaplegic homolog 3 (SMAD3) (4) Protein kinase domain (56-340) in Glycogen synthase kinase-3 beta (GSK3B) Interaction Regulation PTM-dependent Induction (Phosphorylation of S208 on Mothers against decapentaplegic homolog 3 (SMAD3)) of the Mothers against decapentaplegic homolog 3 (SMAD3) MOD_GSK3_1 motif - Glycogen synthase kinase-3 beta (GSK3B) Protein kinase domain interaction Regulatory Enzymes for switch Modifying enzymes for residue: S208: CDC2/CDKX subfamily 2Interaction #3 SMAD3 - NEDD4L Requires Interaction #2 SMAD3 - GSK3B Interfaces (5) ELM:LIG_WW_Nedd4L motif (203GSPNLSPN210) in Mothers against decapentaplegic homolog 3 (SMAD3) (6) WW domain (499-528) in E3 ubiquitin-protein ligase NEDD4-like (NEDD4L) Interaction Regulation PTM-dependent Induction (Phosphorylation of S204 and S208 on Mothers against decapentaplegic homolog 3 (SMAD3)) of the Mothers against decapentaplegic homolog 3 (SMAD3) LIG_WW_Nedd4L motif - E3 ubiquitin-protein ligase NEDD4-like (NEDD4L) WW domain interaction Regulatory Enzymes for switch Modifying enzymes for residue: S204: Glycogen synthase kinase-3 beta (GSK3B); S208: CDC2/CDKX subfamily Additional Information Affinity : T179: 60 µM, pT179: 8.5 µM, pT179-S204-pS208: 2.9 µM, pT179-pS204-pS208: 0.7 µM Structural information: 2LAJ 3Interaction #4 SMAD3 - NEDD4L Requires Interaction #3 SMAD3 - NEDD4L Is mutually exclusive with Interaction #1 SMAD3 - PIN1 Interfaces (7) LIG_WW_1 motif (181PPGY184) in Mothers against decapentaplegic homolog 3 (SMAD3) (8) WW domain (387-416) in E3 ubiquitin-protein ligase NEDD4-like (NEDD4L) Interaction Regulation PTM-dependent Enhancement (Phosphorylation of T179 on Mothers against decapentaplegic homolog 3 (SMAD3)) of the Mothers against decapentaplegic homolog 3 (SMAD3) LIG_WW_1 motif - E3 ubiquitin-protein ligase NEDD4-like (NEDD4L) WW domain interaction Regulatory Enzymes for switch Modifying enzymes for residue: T179: CDC2/CDKX subfamily Inferred Regulatory Enzymes for switch Putative modifying enzymes for residue: T179 : Cyclin-dependent kinase 2 (CDK2), Cyclin-dependent kinase 2 (Cdk2), Cyclin-dependent kinase 4 (Cdk4), Cyclin-dependent kinase 8 (CDK8), Cyclin-dependent kinase 9 (CDK9), Cyclin-dependent kinase 4 (CDK4), Mitogen-activated protein kinase 1 (MAPK1). Additional Information Affinity : T179: 60 µM, pT179: 8.5 µM, pT179-S204-pS208: 2.9 µM, pT179-pS204-pS208: 0.7 µM Structural information: 2LB2 Context Switch localisation ELM curated: nucleus, cytosol, integral membrane protein, integral plasma membrane protein Switch pathways References (1) A Smad action turnover switch operated by WW domain readers of a phosphoserine code. |
|




